CIED Infection Toolkit
Bridging Gaps in Awareness, Detection and Appropriate Treatment of CIED Infections
As many as 1 in 20 patients with a cardiac implantable electronic device (CIED) develop an infection within three years of implantation. Gaps and delays in recommended care for CIED infections are all too common and can lead to preventable illness, disability, and death. The American Heart Association's National CIED Infection Initiative seeks to address the gaps in awareness, detection, and appropriate treatment of cardiovascular implantable electronic device infections.
Use this CIED Infection Toolkit as a resource both to learn about CIED infections and to increase awareness of the public health burden of CIED infection and urgency around timely infection identification and evidence-based treatment. A PDF containing the same information can be downloaded below.
Key Things to Know About CIED Infection
- Cardiac implantable electronic devices (CIEDs), such as pacemakers and implantable cardioverter-defibrillators, save and extend lives with minimal problems in most cases. However, for patients who experience infections related to their devices, gaps and delays in guideline-recommended care can lead to preventable illness, disability and death.
- The presence of abandoned leads is a significant risk factor for infection. These wires, which connect the device to the patient’s heart, are sometimes not removed when the patient receives new leads. Infections are more likely when leads are not properly extracted.
- There are two types of infection: localized (pocket) infection and systemic infection. A localized infection occurs where the device is implanted (pocket) and a systemic infection starts in pocket and spreads to the device leads, the blood stream and can infect the heart.
- Patients with a CIED infection should be referred to a specialist with expertise in device extraction, and the CIED and all its components should be removed. This recommendation is supported by the American Heart Association, Heart Rhythm Society, British Heart Rhythm Society, European Society of Cardiology, and European Heart Rhythm Association. But despite the recommendations, many patients with CIED infections do not undergo complete system removal.
- Patients, caregivers, physicians and other clinicians all play a role in CIED infection care, and communication between these stakeholders is critical. Patient education needs to be based on patient needs and concerns rather than limited to information and instructions.
CIED Infection Overview
Review this video and the content below for an overview on CIED infections.
The use of cardiac implantable electronic devices (CIEDs), such as pacemakers, implantable cardioverter-defibrillators (ICDs), and other implantable devices, is becoming more and more common. While these devices extend and improve people’s lives with minimal problems in most cases, for patients who experience infections related to their devices, gaps and delays in guideline-recommended care can lead to preventable illness, disability and death. Data have shown that these kinds of gaps and delays in guideline-recommended care are all too common1. While so many people have CIEDs, these infections are not rare events; one study found a significant increase in the annual rate of CIED infection from 1.53% in 1993 to 2.41% in 20082. Improved awareness and timely diagnosis of CIED infections are essential to help save lives.
The science is clear about what to do: Patients with a CIED infection should be referred to a specialist with expertise in device extraction, and the CIED and all its components should be removed.
The American Heart Association (AHA) launched an initiative to improve awareness, detection, diagnosis and treatment of CIED infection through a National CIED Infection Summit and Health Care Professional Education(link opens in new window)(link opens in new window)(link opens in new window). For more information about the initiative, please visit: heart.org/treat2beatciedinfection(link opens in new window)(link opens in new window)(link opens in new window).
Types of CIED Infections
Review this video and the content below to learn about the different types of CIED infections.
CIED infections can be categorized as localized to the CIED pocket, such as generator erosion or pocket infection, or as systemic, such as bacteremia or lead-associated endocarditis, an inflammation of the heart lining associated with the leads, or wires, that connect the device to the heart. Local infections can lead to systemic infections if not identified and treated promptly according to established guidelines.
Infection presentation may range from subtle to more obvious:
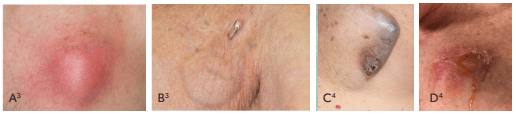
POCKET INFECTION
Pocket infection symptoms may include5 inflammatory skin changes (e.g. pain, swelling or redness), warmth, skin and soft tissue ulceration and drainage, or erosion of generator or protrusion of leads through the skin at the site of the implant pocket.
SYSTEMIC INFECTION
Systemic infection may develop if a localized CIED infection spreads to the device leads, enters the blood stream (bacteremia) or infects the heart (endocarditis). Symptoms of systemic infection may include fever, chills, anorexia, malaise, disorientation, or respiratory distress6.
Evidence Based Diagnostic Treatment Strategies
evidence-based diagnostic and treatment strategies.
Timely detection and diagnosis of a CIED infection is essential for providing best-practice care. Below is a sample CIED infection classification criteria and sample algorithm for suspected CIED Infections.
Pocket Findings*
- Physical exam: device erosion through skin, purulence emanating from pocket, fluctuance, or sinus tract.
- Intraoperative findings: purulence within the generator pocket site.
- Cultures: positive cultures (significant microbial growth, i.e., tissue and swab sample growth when colonies grow on ł2 quadrants of the culture plate and device sonication sample growth when ł20 colonies are isolated from 10 ml of sonicate fluid) from explanted CIED.
Clinical Findings*
MAJOR
- Two or more positive blood cultures for organisms typical of CIED infection, such as S. aureus, coagulase-negative staphylococci (CoNS), or enterococci, with no alternative source.
- TEE findings consistent with vegetation on the device lead and/or heart valve.
- Positron emission tomography-computed tomography (PET-CT) imaging consistent with device infection.
MINOR
- Prolonged bacteremia (>72 h) with microorganisms other than listed in major criteria.
- TEE findings not meeting major criteria.
- Recent pocket manipulation (<3 months prior to presentation).
- Fever (38°C or higher).
- Embolic phenomena (typically septic pulmonary emboli from lead vegetations or right-sided endocarditis).
- Pocket erythema or tenderness.
CIED Infection Classification Criteria*
- Definite CIED infection: combination of any 2 major clinical findings or 1 or more pocket findings.
- Probable CIED infection: 1 major clinical finding and 1 or more minor clinical findings.
- Possible CIED infection: suspected CIED infection case that does not meet “Definite” or “Probable” criteria.
* Proposed MAYO CIED infection classification criteria7
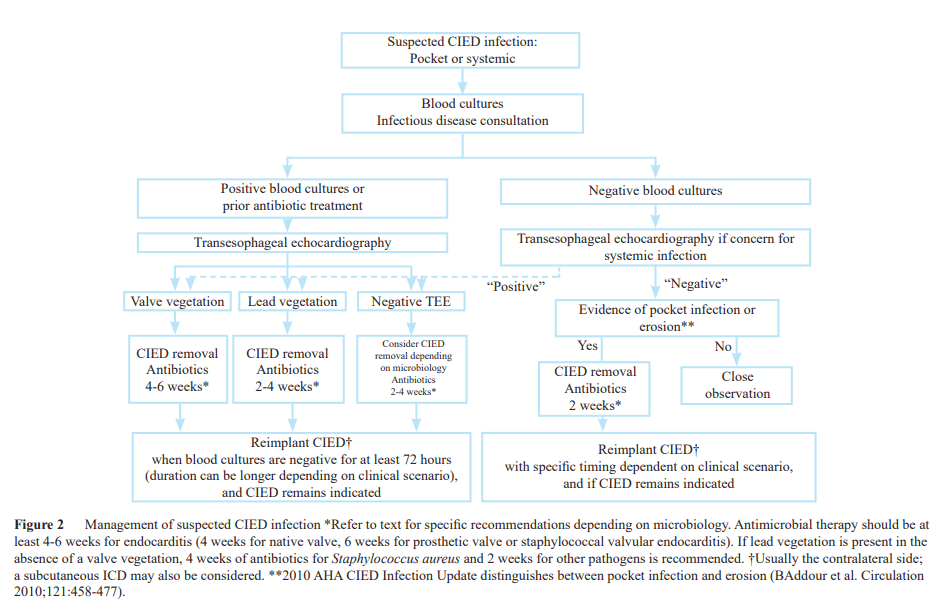
The Heart Rhythm Society’s (HRS) revised 2017 guidelines and 2020 European Heart Rhythm Association’s (EHRA) international consensus document are clear about what to do8,9 :
- Patients presenting with a definite CIED infection, endocarditis (regardless of device involvement), or unexplained or persistent bacteremia or fungemia, should be referred to an expert in the treatment of CIED infection.
- The Heart Rhythm Society’s guidelines also call for antibiotics to be initiated after two sets of positive blood cultures are obtained. However, antibiotic treatment alone is not enough. The device and its components should be removed promptly and completely. Other major professional organizations recommending complete removal in patients with a definite CIED infection include the AHA, British Heart Rhythm Society, and European Society of Cardiology.10,11
Despite the consensus, many patients with CIED infections do not receive recommended care.
Risk Factors
Review this video and the content below to learn about risk factors for CIED infections.
Both the volume and complexity of implantations of pacemakers and other implantable devices has increased over time. The number of implantations increased by 96% between 1993-200812. Additionally, patients having devices implanted are older than before. The risk of infection increases with age, comorbidities such as diabetes and chronic kidney disease, and complexity of the devices13.
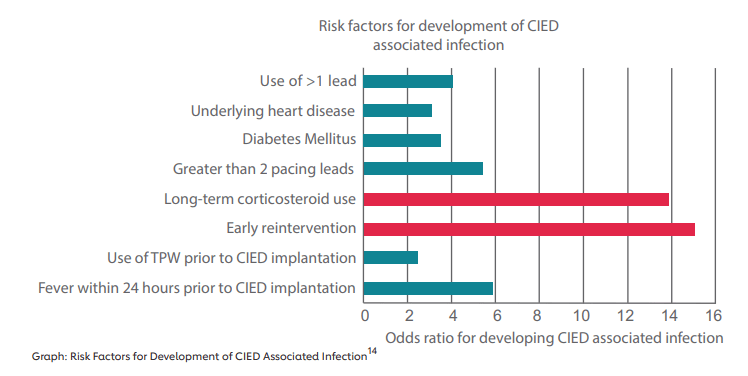
The presence of abandoned leads is a significant risk factor for infection15. These wires, which connect the device to the patient’s heart, are sometimes not removed when the patient receives new leads. Infections are more likely when leads are not properly extracted16. Sometimes, leads are not extracted because physicians and patients think that extraction is riskier than leaving the leads in. However, abandoned leads increase the infection rate, and removing previously abandoned leads from a patient with an infection may carry increased risk. In that situation, patients are more likely to experience a procedural complication when the abandoned leads are extracted17.
Patient comorbidities can also increase the risk of CIED infection17,18:
- diabetes mellitus
- end-stage renal disease
- renal insufficiency
- chronic obstructive pulmonary disease
- malignancy
- heart failure
- pre-procedural fever
- anticoagulant drug use
- skin disorders
- post-operative hematoma
- reintervention for lead dislodgement
- lack of antibiotic prophylaxis
- temporary pacing
- central venous thrombosis in the area of the leads
Financial Burden of CIED Infection
Review this video and the content below to learn
about the financial burden of CIED infections.
Patients and their families bear the greatest cost when CIED infections reduce the quality and length of the patients’ lives. Patients with infections also require additional procedures, and if best-practice management guidelines are not followed, they may experience morbidity and/or mortality as a result. Some costs are direct: Average annual medical costs were 2.4 times higher for CIED patients with an infection, compared to those without an infection19. An analysis of claims through commercial insurers and Medicare supplement insurance estimated that infections increase the per-patient cost of care from $62,256 to $110,141 for initial implants and from $64,810 to $110,332 for replacement implants20. Indirect costs include lost productivity for the patient and family caregivers, and intangibles such as pain, disruption and lost time due to illness, disability and treatment.
Reasons for higher costs:
- High resource utilization
- Hospitalizations
- Cost increased with time to device extraction
- Increased length of stay
Patient Communication Strategies
Review this video and the content below for patient communication strategies.
A group of CIED experts met in March 2022 and identified patient and clinician communication as a common barrier that prevents patients from early infection identification. Patients and physicians play a role in CIED infection care, and communication between these stakeholders is critical. Communicating with your patients about CIED infections or device removal can be a difficult conversation, but a necessary one.
Effective and open communication leaves patients and healthcare teams happier and can result in better health outcomes. To be effective, patient education should be tailored to a patient’s needs and concerns rather than just information and instructions. Here are a few tips to better communicate with your patients 21, 22, 23:
- Be attentive and exercise active listening
- Ask open-ended questions to learn more about your patients
- Involve friends and family
- Use proper tone when communicating with patients to show that you are working together as a team
- Take your patient’s situation and background into account
- Use a multidisciplinary approach
- Be aware of bias
- Communicate in different ways and utilize multimedia – such as short educational videos
- Use shared decision-making
- Keep records and share your notes with patients
- Incorporate “Teach Back” during patient visits to increase understanding 24.
Listen to a conversation about the value of shared decision-making between patient and healthcare professionals when it comes to preventing and managing device infections.(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)
Looking Ahead
Watch this video to learn how you can increase awareness of
CIED infection and promote timely identification and treatment.
This multi-layered call to action relies on healthcare professionals to evaluate how CIED infection patients are being treated, drive guideline adherence and get the message out that gaps in care exist. Patients are called upon to be advocates for their own health. In March 2022, the AHA led by a nine-member planning committee convened multidisciplinary stakeholders at an in-person CIED Infection Summit (link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)that focused on three categories of action:
Driving Detection and Diagnosis: Identifying the most critical problems across clinical settings and connecting the dots for clinicians, including the role of informatics.
Improving Treatment and Management of CIED Infection: Following best practices for enhancing systems of care.
Awareness and Education: Learning from impactful consumer and healthcare professional initiatives in other diseases.
Use this toolkit, recorded webinars, podcasts and other resources made available from AHA’s CIED Infection Initiative(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window) to increase awareness of CIED Infection and evidence-based treatment and management.
Infections are a lifelong risk for patients with CIEDs, but prompt, expert, guideline-directed treatment can reduce the impact of infections on patients’ lives. Quality improvement initiatives and care redesign programs can enhance the care that patients with CIEDs receive within health systems. These initiatives should build greater awareness among patients, caregivers and health care professionals of the risk of infection and the best ways to manage it; promote earlier detection and diagnosis of infection; encourage guideline directed treatment and management; and establish measurement of and feedback on care performance.
Review the CIED Infection Initiative proceedings document(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window)(link opens in new window) for key summit takeaways and preliminary actionable solutions.
CIED Infection Toolkit References
1. Voigt A, Shalaby A, Saba A. Rising rates of cardiac rhythm management device infections in the United States: 1996 through 2003. J. Am. Coll. Cardiol. 2006 Aug 1;48(3):590-591.
2. Greenspon AJ, Patel JD, Lau E, et al. 16-Year trends in the infection burden for pacemakers and implantable cardioverter-defibrillators in the United States: 1993 to 2008. J Am Coll Cardiol. 2011;58:1001-1006.
3. Hui-Chen Han, Nathaniel M Hawkins, Charles M Pearman, David H Birnie, Andrew D Krahn, Epidemiology of cardiac implantable electronic device infections: incidence and risk factors, EP Europace, Volume 23, Issue Supplement_4, June 2021, Pages iv3–iv10.
4. DeSimone DC, Sohail MR, Mulpuru SK. Contemporary management of cardiac implantable electronic device infection. Heart. 2019 Jun;105(12):961-965. doi: 10.1136/heartjnl-2017-312146. Epub 2019 Feb 12. PMID: 30755468.
5. Fakhro A, Jalalabadi F, Brown RH, Izaddoost SA. Treatment of Infected Cardiac Implantable Electronic Devices. Semin Plast Surg. 2016 May;30(2):60-5. doi: 10.1055/s-0036-1580733. PMID: 27152097; PMCID: PMC4856529.
6. Refaat M, Zakka P, Khoury M, Chami H, Mansour S, Harbieh B, Abi-Saleh B, Bizri AR. Cardiac implantable electronic device infections: Observational data from a tertiary care center in Lebanon. Medicine (Baltimore). 2019 Apr;98(16):e14906. doi: 10.1097/MD.0000000000014906. PMID: 31008922; PMCID: PMC6494368. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6494368/.
7. DeSimone DC, Sohail MR. Approach to Diagnosis of Cardiovascular Implantable-Electronic-Device Infection. J Clin Microbiol.2018;56(7):e01683-17. Published 2018 Jun 25. doi:10.1128/JCM.01683-17.
8. Kusumoto FM, Schoenfeld MH, Wilkoff BL, et al. 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction [published correction appears in Heart Rhythm. 2021 Oct;18(10):1814]. Heart Rhythm. 2017;14(12):e503-e551.
9. Blomström-Lundqvist C, Traykov V, Erba PA, et al. European Heart Rhythm Association (EHRA) international consensus document on how to prevent, diagnose, and treat cardiac implantable electronic device infections-endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), the Latin American Heart Rhythm Society (LAHRS), International Society for Cardiovascular Infectious Diseases (ISCVID) and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Europace. 2020;22(4):515-549.
10. Baddour LM, Epstein AE, Erickson CC, et al. Update on cardiovascular implantable electronic device infections and their management: a scientific statement from the American Heart Association. Circulation. 2010;121(3):458-477.
11. Sandoe JA, Barlow G, Chambers JB, et al. Guidelines for the diagnosis, prevention and management of implantable cardiac electronic device infection. Report of a joint Working Party project on behalf of the British Society for Antimicrobial Chemotherapy (BSAC, host organization), British Heart Rhythm Society (BHRS), British Cardiovascular Society (BCS), British Heart Valve Society (BHVS) and British Society for Echocardiography (BSE). J Antimicrob Chemother. 2015;70(2):325-359.
12. Greenspon AJ, Patel JD, Lau E, et al. 16-Year trends in the infection burden for pacemakers and implantable cardioverter-defibrillators in the United States: 1993 to 2008. J Am Coll Cardiol. 2011;58:1001-1006.
13. Patel J, Kurtz S, Lau E, et al. Removal/replacement procedures and deep infection risk for pacemakers and ICDs in the United States: Medicare analysis from 1997 to 2010. Heart Rhythm. 2013;10:S118.
14. David G. Wilson, Arthur Yue, John M. Morgan, Paul R. Roberts, Strategies to reduce infections during the cardiac implantable electronic device implant: a time to name and shame , EP Europace, Volume 17, Issue 4, April 2015, Pages 511–513.
15. Pokorney SD, Mi X, Lewis RK, et al. Outcomes associated with extraction versus capping and abandoning pacing and defibrillator leads. Circulation. 2017 Oct 10;136(15):1387-1395.
16. David G. Wilson, Arthur Yue, John M. Morgan, Paul R. Roberts, Strategies to reduce infections during the cardiac implantable electronic device implant: a time to name and shame , EP Europace, Volume 17, Issue 4, April 2015, Pages 511–513.
17. Tarakji KG, Ellis CR, Defaye P, Kennergren C. Cardiac Implantable Electronic Device Infection in Patients at Risk. Arrhythm Electrophysiol Rev. 2016 May;5(1):65-71. doi: 10.15420/aer.2015.27.2. PMID: 27403296; PMCID: PMC4939310.
18. Konstantinos A. Polyzos, Athanasios A. Konstantelias, Matthew E. Falagas, Risk factors for cardiac implantable electronic device infection: a systematic review and meta-analysis, EP Europace, Volume 17, Issue 5, May 2015, Pages 767–777.
19. Eby EL, Bengtson L, Johnson M, et al. Economic impact of cardiac implantable electronic device infections: cost analysis at one year in a large U.S. health insurer. J Med Econ. 2020 Jul;23(7):698-705.
20. Incidence, treatment intensity, and incremental annual expenditures for patients experiencing a cardiac implantable electronic device infection: evidence from a large US payer database 1-year post implantation. Circ Arrhythm Electrophysiol. 2016 Aug;9(8):e003929.
21. Chan, Siobhan. “16 ways to improve your communication skills with patients” British Heart Foundation. Jan 9 2020.
22. MedlinePlus [Internet]. Dugdale (MD), et al: National Library of Medicine (US); [updated 2021 Oct 27]. Communicating with patients. Available from: https://medlineplus.gov/ency/patientinstructions/000456.htm(link opens in new window).
23. King A, Hoppe RB. “Best practice” for patient-centered communication: a narrative review. J Grad Med Educ. 2013 Sep;5(3):385-93. doi: 10.4300/JGME-D-13-00072.1. PMID: 24404300; PMCID: PMC3771166.
24. Teach-Back Method: Tool #5. Content last reviewed September 2020. Agency for Healthcare Research and Quality, Rockville, MD. https://www.ahrq.gov/health-literacy/improve/precautions/tool5.html.

Sponsor
Philips Image-Guided Therapy is a proud supporter of the American Heart Association's National CIED Infection Initiative.